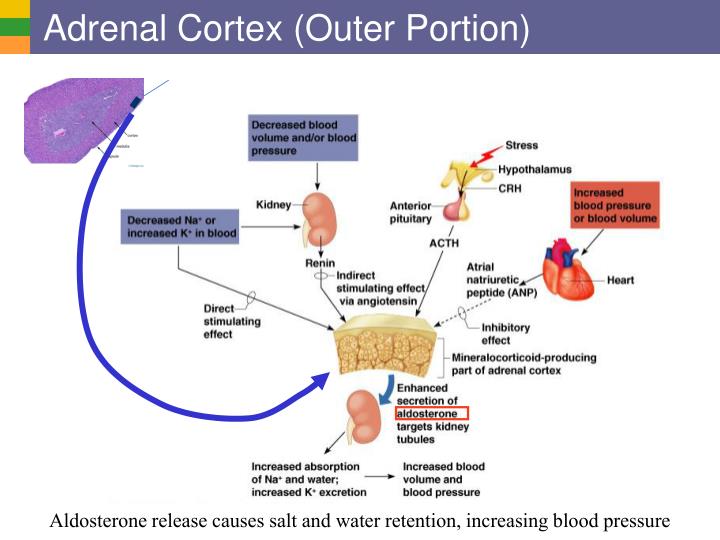
Loss of feedback inhibition was compensated by increased tyrosine hydroxylase activity, as detected by elevated tissue dihydroxyphenylalanine levels. In vivo, deletion of sympathetic or adrenal feedback control led to increased plasma and urine norepinephrine (α 2A-knockout) and epinephrine levels (α 2C-knockout), respectively. The inhibitory effects of α 2-agonists on cell capacitance, voltage-activated Ca 2+ currents, and on catecholamine secretion were completely abolished in chromaffin cells isolated from α 2C-receptor-deficient mice.  
In isolated mouse chromaffin cells, α 2-receptor activation inhibited the electrically stimulated increase in cell capacitance (a correlate of exocytosis), voltage-activated Ca 2+ current, as well as secretion of epinephrine and norepinephrine. Using gene-targeted mice we demonstrate that two distinct subtypes of α 2-adrenoceptors control release of catecholamines from sympathetic nerves (α 2A) and from the adrenal medulla (α 2C). In the adrenergic system, release of the neurotransmitter norepinephrine from sympathetic nerves is regulated by presynaptic inhibitory α 2-adrenoceptors, but it is unknown whether release of epinephrine from the adrenal gland is controlled by a similar short feedback loop.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
